Reverse osmosis is a method used to separate a high percentage of pollutants from water by forcing it through a semi-permeable membrane.
Issues regarding freshwater production have persisted throughout human history. Several factors such as location, contaminants, temperature, salinity, dissolved solids, and others have hampered the distribution of clean water in many regions. Fortunately, the introduction of reverse osmosis has provided an efficient solution to this problem. This article is intended for those who do not have a clear understanding with reverse osmosis water and will illustrate the basics of reverse osmosis technology and its applications.
Reverse osmosis operates by utilizing advanced filtration technology which removes dissolved ions from water. Osmosis is an elemental force that draws water to water with higher salt content. This is a process by which dissolved ions are removed from water. This elemental force can be surmounted by applied pressure with the usage of pumps and semi-permeable membranes, which forces water through the membrane and filters out dissolved salt from the water.
- Understanding the Definition of Osmosis and Reverse Osmosis Water?
- How does a Reverse Osmosis System work?
- What are the Basic Components of a Reverse Osmosis System?
- What Types of Commercial/Industrial Applications do RO systems Serve?
- What Types of Water Sources Does Reverse Osmosis Treat?
- Is Pretreatment Necessary?
- Is it Necessary to get a Water Analysis?
- What Determines the Precise Pretreatments for a Particular RO?
- Does the Feed Water need to be Softened by the RO?
- What is Dechlorination used for?
- Is Acid Injection Necessary?
- What does Antiscalant do?
- Do all Reverse Osmosis Systems Require Chemical Cleaning?
- Summary
- Request a quote
Reverse osmosis is a hot topic in the water treatment industry. With the lowest energy requirements, some of the highest recovery rates, and one of the best rejection rates on the market, it's no wonder people are interested in learning more about it. What is the definition of reverse osmosis, though? How does it work? Let's take a look into the heart of a Pure Aqua RO system and break it down for you to understand all about RO.
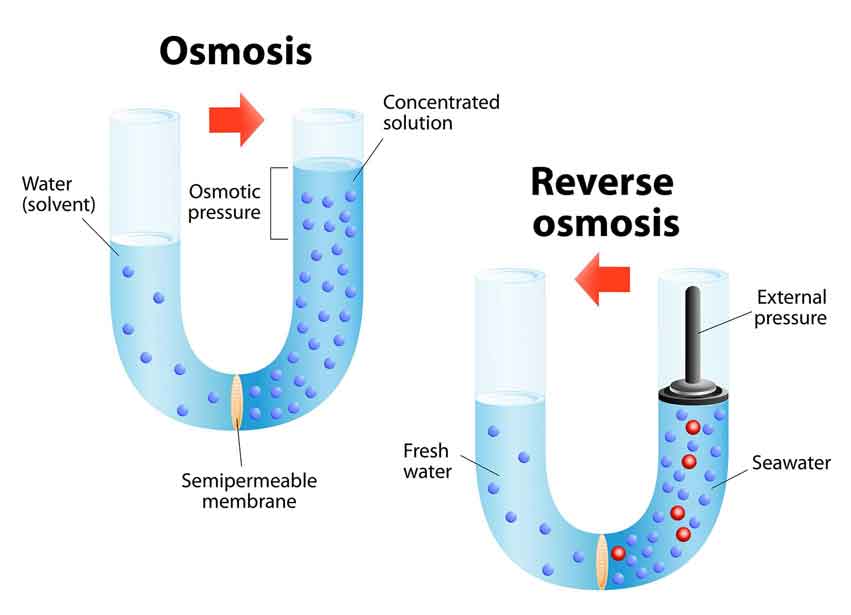
As anyone could guess, it is the process of osmosis backwards. Osmosis is the passage of water through a reverse osmosis membrane (like our skin, or the inside of a plant cell) to equalize the concentration of particles dissolved in the water. As the diagram above shows, the semi-permeable membrane allows water to pass through, but molecules larger than water (things like minerals, salts, and bacteria) cannot. Water flows back and forth until the concentration is equal on both sides of the membrane, and an equilibrium is formed.
Let's apply this knowledge to water purification. We want to drink water from a lake or stream, but it contains too high concentration of contaminants like salt, minerals, and bacteria, that make it undrinkable. By applying pressure to water as it passes through a membrane, the water can be forced to move away from the membrane rather than attempting to form an equilibrium like normal. This against-flow motion is where the "reverse" in "reverse osmosis" comes from. A pump works well for this process. Water is forced through the membrane, which like a super-fine particle filter, blocks an extreme majority of contaminants from coming through.
As a water purification process, it has a number of advantages as well as disadvantages. In water treatment, TFC membranes can usually remove between 96 and 99% of most contaminants, including salts and minerals, dyes, particles, bacteria, and hazardous metals. Because of the way reverse osmosis works, however, you can never truly remove all of a contaminant. You may purify down to a fraction of a fraction of a percent, but the contaminant can never be truly eliminated with reverse osmosis. Water purification systems also requires a superior quality, high pressure pump, because the rejection rate is primarily dependent on the pressure applied to the membrane. That being said, smaller units have smaller ratios of permeate (clean, purified product) to waste water. This makes media filtration or other conventional filtration more effective in smaller scales (like residential settings).
Now that we've answered the biggest question about membranes systems by Pure Aqua, let's take a closer look at how we build our advanced water treatment systems and how reverse osmosis works.
Reverse Osmosis works by filtering unwanted pollutants such as bacteria, viruses and other microbiological organisms from water by pressuring the treated water which forces water substances through a semipermeable membrane. Throughout the process, the pollutants are separated from the water and flushed out, which produces ultra pure water.
The reverse osmosis process works by utilizing high pressure pumps to elevate the pressure on the concentrated side of the RO system and push the water through membrane(s). It is a highly effective water treatment process that needs a higher degree of pressure depending on the total amount of contaminants existing in the feed water.
The purified water that is removed of its dissolved solids is now referred to as permeate or product water. The concentrated stream of contaminants left behind in the brine is referred to as reject water and is ultimately discharged.
The reverse osmosis membranes sole responsibility is to ensure efficient filtration of all unwanted salts and minerals from the feed water as it passes through the system. Membranes from top-branded manufacturers (Dupont Filmtec, Hydranautics) are capable of eliminating up to 99% of dissolved solids from the water source. The rejected stream is not always thrown away, it can be recycled through the reverse osmosis system for water conservation.
The reverse osmosis process is more similar to a cross-flow filtration method than a conventional one. Cross-flow filtration is more effective since it consists of two outlets for the solution to go in order to handle higher concentrations of pollutants. The flow of the permeate and concentrated water in opposite directions allows the membrane surface to remain clean and greatly minimizes buildup. This filtration method ensures a longer-life span of the system and components and reduced the need for membrane cleaning.
Now that we know how reverse osmosis works, let's take that and apply it to a real, working TWRO or BWRO system. If it only needed the membranes and a pump, it certainly wouldn't be so big, right?
Here is the complete reverse osmosis process, including the pretreatment and post-treatment that is generally required for highly contaminated water:
A) Pre-Chlorination Dosing System
If the feed water contains traces of heavy metals or contaminated, it is highly recommended to dose some chlorine to change the dissolved heavy metals to physical form, the media filter will be able to filter most of it.
B) Raw Water Storage Tank
Although some RO Systems can draw water right out of a well or pipe feed, most reverse osmosis systems start with a large tank that stores the contaminated water. Not having enough feed water can damage a pump, so having a large storage tank for your intake water is an easy way to make sure your pump lasts for as long as possible.
C) Feed Water Pump
A commercial or industrial pump provides the initial pressure for the Treatment System. This motor usually provides enough water pressure to get through any pretreatment as well as the RO membranes, but if it doesn't a booster pump may be necessary farther down the line.
D) Multi-Layer or Media Filter
As much as we hate to admit it, there are some things that membranes can't purify. Nitrates, a common contaminant found in fertilizers and animal waste, are a good example of particles that dissolve too well in water for reverse osmosis to catch them. Things like foul odor and taste usually aren't prevented by reverse osmosis, either. A Multi-Layer filter can be filled with media that specifically targets the things your RO system can't catch. If you need to eliminate these contaminants, a multi-layer filter is a must. An example of MMF or multi media filters is our water media filters MF-1000 Series.
E) Activated Carbon Filter
Activated carbon filters are a good solution to reduce organic, bad taste, smell and chlorine from the water.
F) Automatic Water Softener
Automatic water softeners are designed to remove water hardness, calcium and magnesium ions, for small RO systems, we usually recommend water softeners instead of the antiscalant chemical dosing.
G) Antiscalant Chemical Dosing System
For larger RO systems, we use antiscalant dosing systems to dose our PA0100 antiscalant RO chemical, which helps in preventing membrane fouling. For more information, please visit our CDS-Series chemical dosing pumps.
H) Reverse Osmosis System
We finally have our reverse osmosis system. If a booster pump is necessary, it will typically be just before this step. The reverse osmosis system can produce up to one million gallons of product water a day from a steady intake, as well as a sizable amount of waste. Usually the wastewater can be dumped down the drain, but check with your local water authorities in case it needs to be handled with care.
I) Product Water Storage Tank
The permeate from the reverse osmosis system will usually go to a large tank, where it is held for use. If it didn't, the system would need to be running in order to have access to fresh water, which can be inconvenient. Sometimes, a RO system pumps water directly into a well or aquifer for recharging instead of being used in many of the normal industries or applications it is used in.
J) Post-Chlorination Dosing System
If the permeate water is intended to be stored for more than one day, it is highly recommended to dose some chlorine to maintain clean and non contaminated water.
K) Product Water Pump (Re-pressurization)
This pump repressurizes the permeate water to the point of use ends. This is selected based on the overall travelling distance and required head. This pump must be selected in stainless steel to prevent any contamination to the permeate water.
L) Product Water UV Sterilizer
The UV sterilizer is placed after the storage tank, and as a final disinfection device. Most of the time, we either use post chlorination as a disinfectant agent, or ultraviolet sterilization.
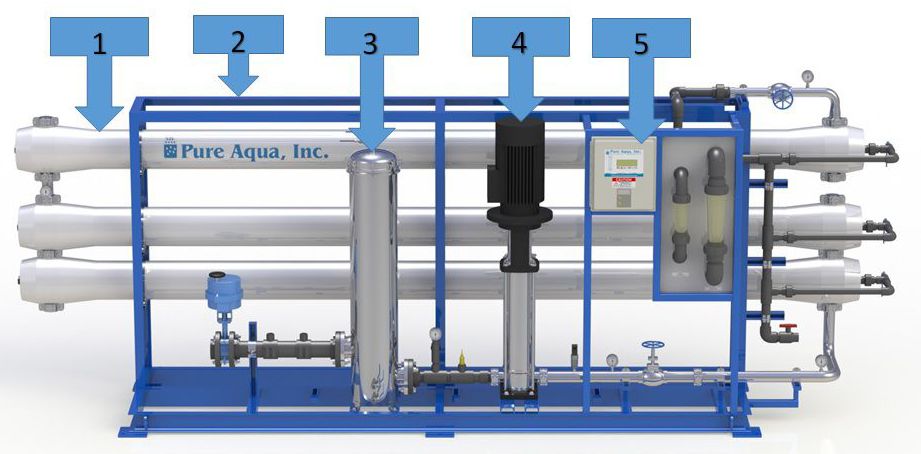
A Reverse Osmosis System is built of five basic parts:
1) Pressure Vessels & Membranes
Obviously, a reverse osmosis system wouldn't get very far without membrane elements. The proteins that make up membrane elements vary depending on the kind of intake water and ending clarity. There are membrane elements for brackish water, seawater, hospital-grade disinfection, and membranes designed to remove specific contaminants to name a few. If there is a water treatment need, you can be sure there is certainly a membrane element for the job. The size of the task (municipal, commercial, or industrial) will determine the size and number of reverse osmosis membranes in a system. There can be anywhere from a single two and a half inch membrane (like in an under-sink reverse osmosis water system) to hundreds of eight inch membranes all working together (a typical reverse osmosis plant).
2) Reverse Osmosis Skid
The best way to make your reverse osmosis system as durable as possible is with a powder coated, carbon steel frame to mount all of your components on. It's resistant to the elements, designed for the heavy vibration of high-pressure pumps, and mounts to the ground to ensure that it will last a lifetime.
3) Cartridge Filter
Most reverse osmosis water systems come with a cartridge filter to ensure that no particles large enough to damage the membranes come anywhere near them. This cartridge is usually a five micron spun polypropylene filter, but can vary upon request. The cartridge comes in a durable casing that can handle the pressure from the main feed or booster pumps.
4) Reverse Osmosis High Pressure Pump
Without a high-grade pump, the rejection rate for a reverse osmosis system isn't viable in most commercial or industrial settings. It is vital to the system to ensure that the pump is matched to the membrane quantity and size appropriately. Usually, the higher the horsepower on the intake pump, the better the rejection and recovery rates of permeate.
5) Control Panel
Lastly, the reverse osmosis system has to be controlled by a human operator. At Pure Aqua, we use advanced PLCs or a solid-state microprocessor depending on how advanced the controls need to be. The controls can also be used to manage multiple systems simultaneously, effectively making a one-man water production plant.
A reverse osmosis system can have a number of other components built onto or into it as extra components as well. The entire skid can be built into a containerized system, for example, so your Desalination by RO System is always on the go. There are a number of supplemental skids that can be attached to an RO System as well, for tasks like membrane cleaning, pretreatment, chemical dosing, and a number of other jobs as necessary.
If there is a necessity for water treatment, chances are a reverse osmosis system can do the job. There's a wide array of industries that benefit from having highly pure reverse osmosis water, as well as a large number of applications where water treatment is required. Because of the extreme volumes of water required, a reverse osmosis system is often the ideal, economical solution, requiring less energy than most large-scale treatment methods. Because they consume less energy, a reverse osmosis system is often the environmentally friendly solution as well. At Pure Aqua, we pride ourselves as being a go-to source for information and service helping you solve your municipal, commercial, or industrial water treatment needs.
- Pretreatment of boilers
- Food and beverage treatment
- Industrial wastewater purification
- Deionized water filters
- Hotels and resorts
- Ice making
- Spot free car washes
- Pure ethanol
- Dairy industry
- Maple syrup
- Pharmaceuticals
- Water bottling
- Hospitals
- Agriculture
- Humidification
Reverse Osmosis is an ideal water treatment solution in most types of water. Generally speaking, all major water sources from a treatment standpoint can be broken down into three major categories: tap water, also known as municipal sources, groundwater, which includes brackish water, and saltwater. The biggest distinction between these three types is the Total Dissolved Solids (TDS) content of each type. As a rule of thumb, the American Health Association requires that drinking water is under 1,000 PPM TDS.
Tap water typically comes through a pre-existing infrastructure like city pipes or a damming system. Reverse osmosis is often used in a tap water environment to reduce hardness, or the debris deposited in water from traveling in metal pipes. Total dissolved solids is often a target of water purification in tap water systems. RO water is ideal in applications like power plants, pharmaceuticals, laboratories, and hospitals, where an extreme purity of water is crucial to the industry. Tap water typically has a TDS of under 1,000 PPM.
Underground reservoirs of water are often brackish or highly brackish, meaning they contain large volumes of salt, but not enough to be considered salt water. Groundwater reverse osmosis is very common, and one of the best uses of a reverse osmosis system to date. Groundwater is most often purified for the agriculture industry, the mining industry, and for residential use. Groundwater is also a prized target of the bottling industry, because the unique mineral combinations often have an appealing taste. Brackish water usually has a TDS of 5,000 PPM or less, but can come in concentrations of up to 12,000 PPM.
Reverse osmosis salt water (sometimes referred to simply as desalination) is the turning of salt water into drinking water. Ocean water has up to 45,000 PPM TDS. Typically, for environmental reasons, a bore hole is dug in the ocean for this kind of reverse osmosis water, but an open intake is more cost effective. The biggest uses of desalination come in providing water in areas that lack a regular supply of fresh water.
If you work with a reverse osmosis system, you understand that the feed water must be preconditioned to protect the membranes from fouling and premature failure. An RO membrane functions much like a cross flow filter. The membrane is constructed of a porous material that allows water to pass through the membrane, but rejects up to 99% of the dissolved solids at the membrane surface. The dissolved salts are concentrated in the reverse osmosis reject water, or brine stream, where they are discharged to waste.
As the RO system continues to operate, the dissolved and suspended solids in the feed water tend to accumulate along the membrane surface. If these solids are allowed to build up, they eventually restrict the passage of the RO water through the membrane, resulting in a loss of throughput. (The throughput capacity of the membranes is commonly referred to as the flux rate, and is measured in gallons per square foot of membrane surface area per day.)
Early in the development of membrane systems, little was known about which impurities in the reverse osmosis feed water are likely to cause fouling and a corresponding reduction in flux. Today, many of these troublesome impurity treatments have been identified, and preventive treatments have been devised that greatly reduce membranes fouling, thus prolonging the life of the RO plant.
Autopsies of failed membranes modules have revealed a build-up of foulants caused by mineral scales such as calcium carbonate; colloidal materials like clays and silica; dead and living microorganisms; carbon particles; and chemical attached by oxidizing agents like chlorine, ozone, or permanganate. Likewise, dissolved metals like iron and aluminum, whether naturally occurring or added as a coagulant, can cause premature fouling and failure of the reverse osmosis membrane.
A detailed chemical analysis of the RO feed water is an absolute necessity for identifying potential foulants. This should include a measurement of the hardness (calcium and magnesium), barium, strontium, alkalinity, pH, and chlorine. The data from the chemical analysis can be used by the RO equipment designers to determine the optimum membrane array that will both minimize the tendency of scale and deposit formation and maximize the recovery and flux rate.
For example, the Langelier Stability Index (LSI), a measure of the calcium carbonate scaling tendency of the water, is computed from the water analysis to determine the maximum permissible concentration of dissolved minerals in the reject stream before scale deposition becomes a problem. Because of the number of variables that must be considered, these calculations are difficult to do with pencil and paper. Fortunately, the membranes manufacturers have developed computer programs that make these computations fast and easy to perform where the user can project the performance of membranes at actual feed conditions.
Although a water analysis is helpful in predicting the tendency of dissolved minerals to cause problems in the RO System, it does not always forecast the fouling tendency of colloids and other finely dispersed suspended solids. The Silt Density Index (SDI) is a useful tool for quantifying the fouling tendency of the feed water. This test is conducted by filtering a sample through a 0.45 micron (µm) filter and measuring the time required to collect a unit volume of filtrate. An index number is calculated from this data. Traditionally, a SDI value of less than 3.0 is desirable for reverse osmosis feed waters. The SDI measurement has certain limitations in that it does not model the cross flow design of an RO membrane.
In one word: analysis. Every source of water is different, and you never know what's in your water until you have it analyzed. The water analysis, LSI, SDI, or CFI values are used to determine the precise pretreatment requirements for a particular RO System. Since water supplies vary considerably from one location to another, each pretreatment requirement will be different. On average, most Reverse osmosis water systems need an antiscalant injector or water softener to prevent damage to the RO membrane.
Ion exchange is a popular method for reducing the potential for mineral scale formation on the membrane surface. Ion exchange softening uses sodium to replace scale-forming ions such as calcium, magnesium, barium, strontium, iron, and aluminum to prevent damage to the membrane elements. The sodium forms very soluble salts, which are readily rejected by the reverse osmosis system and do not readily form mineral scales on the membrane surface. A sodium-cycle softener is regenerated with sodium chloride brine. The spent regenerant, along with the softener rinse water, must be discharged to waste. It is due to this process that ion exchange is recommended for applications that have high metal contents in the treated water.
Generally speaking, chlorination is a double-edged sword when it comes to reverse osmosis systems. As a method of disinfection, chlorination is not only efficient and practical, but it is cost-effective as well. The only problem is that chlorine is too caustic for RO membrane elements, and can cause serious damage. Dechlorination is a kind of chemical injection that adds a chemical that forms salts with chlorine, making it readily rejected by the membrane elements. In this circumstance, dechlorination is a must as far as water treatment is concerned. Without dechlorination, reverse osmosis membranes are not only ineffective when it comes to chlorinated water, but the chlorine will simply destroy the protein membrane.
Just as acidic solutions aren't good for membranes, caustic solutions are equally damaging to reverse osmosis membrane elements. Acid injection may be incorporated into the RO pretreatment system to control pH and minimize the scale-forming tendency of the feed water. Acid injection is indicated if the scale-forming tendency of the brine stream is above +0.3 as measured by the LSI. Either sulfuric or hydrochloric acid can be used for this purpose. However, sulfuric acid is less costly, and is more commonly used.
Antiscalants have been shown to be effective in extending the intervals between chemical cleanings of the RO membranes. These products are generally formulated to include inorganic phosphates, organophosphonates, and dispersants. Use antiscalant products that have been approved by the membrane manufacturer, and follow all directions in applying and controlling the product dosage. Some antiscalant contain negatively charged polymers and dispersants that can react with cationic polymers that might be dosed up stream prior to the media filters. The antiscalant must be compatible with these polymers; otherwise, the reaction product will foul the membranes.
Despite all efforts to protect the RO system from fouling and loss of flux, eventually the membranes will require chemical cleaning. A well-designed RO system will include provisions for a cleaning skid to facilitate the cleaning process. The skid should include a chemical tank, solution heater, recirculating pump, drains, hoses, and all other connection and fittings required accomplishing a complete chemical cleaning of the RO modules.
Various chemical cleaning agents are available for maintaining RO membranes. The type and amount of foulant will dictate the most effective cleaning agent. Acid cleaners’ best remove mineral scale deposits. Hydrogen peroxide is commonly used to clean and sanitize membranes to correct or prevent biofouling problems. In some cases, a mild solvent such as methanol is used. Because of the number of variables involved in the selection and application of these cleaning agents, contact the membrane manufacturer, equipment supplier, or a qualified chemical consultant for specific advice and recommendations on how to accomplish an effective cleaning.
The operation of the RO System should be carefully monitored to predict when the membranes would require cleaning. As a rule of thumb, cleaning is indicated when the normalized flux rate decreases by 10%. Under ideal condition, assuming that the RO pretreatment system is properly designed and operated, the frequency between membrane cleanings should be 3 months or more. Cleaning every 1 to 3 months is considered a fair performance, and suggests that some improvements in the pretreatment system should be considered. Cleaning frequencies every month or more indicate a change in raw water quality, a problem with the pretreatment system, or a problem with the operation of the RO unit.
Reverse osmosis is a reliable method for producing high-purity water. However, most water supplies require some form of RO pretreatment such as softening, media filtration, activated carbon, or chemical injection to protect the reverse osmosis membranes from premature fouling or failure. The pretreatment requirements will vary from location to location, but the overall objective remains the same: to maintain the design flux rates, minimize the membranes cleaning frequency, and prolong the useful life of the RO equipment.
Pure Aqua has over 20 years of experience as a global provider of B2B water treatment solutions for a variety of applications and industries, we offer a large selection of all types of reverse osmosis and water treatment systems to meet your industrial needs. Pure Aqua’s extensive global experience in engineering and manufacturing allows us to pre-engineer and customize water treatment and reverse osmosis systems to meet a wide range of customer requirements and specifications.
 ENGLISH
ENGLISH
 ESPAÑOL
ESPAÑOL العربية
العربية PORTUGUÉS
PORTUGUÉS FRANÇAIS
FRANÇAIS